Chapter 8 Vocabulary
Alloy- The mixture of elements that has metallic properties;most commonly forms when the elements are either similar in size (substitutional alloy) or the atoms of one element are much smaller that the atoms of the other (interstitial alloy).
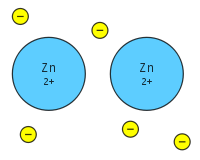
Anion- An ion that has a negative charge; forms when valence electrons are added to the outer energy level, giving the ion a stable electron configuration.
Cation- An ion that had a positive charge; forms when valence electrons are removed, giving the ion a stable electron configuration.
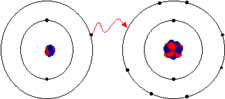
Chemical Bond- The force that holds two atoms together' may form by the attraction of a positive ion for a negative ion or by the attraction of a positive nucleus for negative electrons.
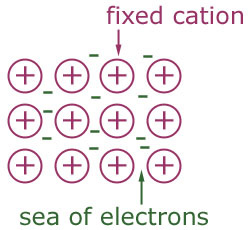
Delocalized Electrons- The electrons involved in metallic bonding that are free to move easily from one atom to the next throughout the metal and are not attached to a particular atom.
Electrolyte- The ionic compound whose aqueous solution conducts an electric current.
Electron Sea Model- Proposes the all metallic solids contribute their valence electrons to form a "sea" of electrons, and can explain properties of metallic solids such as malleability, conduction and ductility.
Formula Unit- The simplest ratio of ions represented in the ionic compound.
Ionic Bond- The electrostatic force that holds oppositely charged particles together in an ionic compound.
Lattice Energy- The energy required the separate one mole of the ions of an ionic compound, which is directly related to the size of the ions bonded and is also affected by the charge of the ions .
Metallic Bond- The attraction of a metallic cation for delocalized electrons.
Monatomic Ion- An ion formed form only one atom.
Oxidation Number- The positive or negative charge of an monatomic atom.
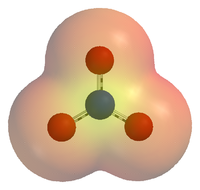
Oxyanion- Polyatomic ion composed of of an element, usually a nonmetal, bonded to one or more oxygen atoms.
Polyatomic Ion- An ion made up of two or more atoms bonded together that acts as a single unit with a net charge.
Anion- An ion that has a negative charge; forms when valence electrons are added to the outer energy level, giving the ion a stable electron configuration.
Cation- An ion that had a positive charge; forms when valence electrons are removed, giving the ion a stable electron configuration.
Chemical Bond- The force that holds two atoms together' may form by the attraction of a positive ion for a negative ion or by the attraction of a positive nucleus for negative electrons.
Delocalized Electrons- The electrons involved in metallic bonding that are free to move easily from one atom to the next throughout the metal and are not attached to a particular atom.
Electrolyte- The ionic compound whose aqueous solution conducts an electric current.
Electron Sea Model- Proposes the all metallic solids contribute their valence electrons to form a "sea" of electrons, and can explain properties of metallic solids such as malleability, conduction and ductility.
Formula Unit- The simplest ratio of ions represented in the ionic compound.
Ionic Bond- The electrostatic force that holds oppositely charged particles together in an ionic compound.
Lattice Energy- The energy required the separate one mole of the ions of an ionic compound, which is directly related to the size of the ions bonded and is also affected by the charge of the ions .
Metallic Bond- The attraction of a metallic cation for delocalized electrons.
Monatomic Ion- An ion formed form only one atom.
Oxidation Number- The positive or negative charge of an monatomic atom.
Oxyanion- Polyatomic ion composed of of an element, usually a nonmetal, bonded to one or more oxygen atoms.
Polyatomic Ion- An ion made up of two or more atoms bonded together that acts as a single unit with a net charge.